A guide to AbSolution
AbSolution is an interactive tool for exploring immune
repertoires and their sequence-based features. AbSolution
has been designed under the principles of accessibility, scalability,
flexibility, interactivity and reproducibility to be approachable to all
users while customizable for the different AIRR-Seq studies.
This vignette offers an overview of AbSolution’s
functionality, guides you on how to initialize the app with your own
data, and how to customize the app.
Launching AbSolution
This is easy. Only two lines are required.
# Load required package
library(AbSolution)
# Load Shiny app
AbSolution::run_app()Make sure you have space in your hard-drive! It can very easily occupy quite a lot of gigabytes.
Quick Help
Need customized information about your current step?
Check the Help subtab in the AbSolution
tab.
Looking for an in-depth explanation of an option? Activate tooltip mode by clicking the “?” at the top right corner to see explanations for each option when you hover over them.
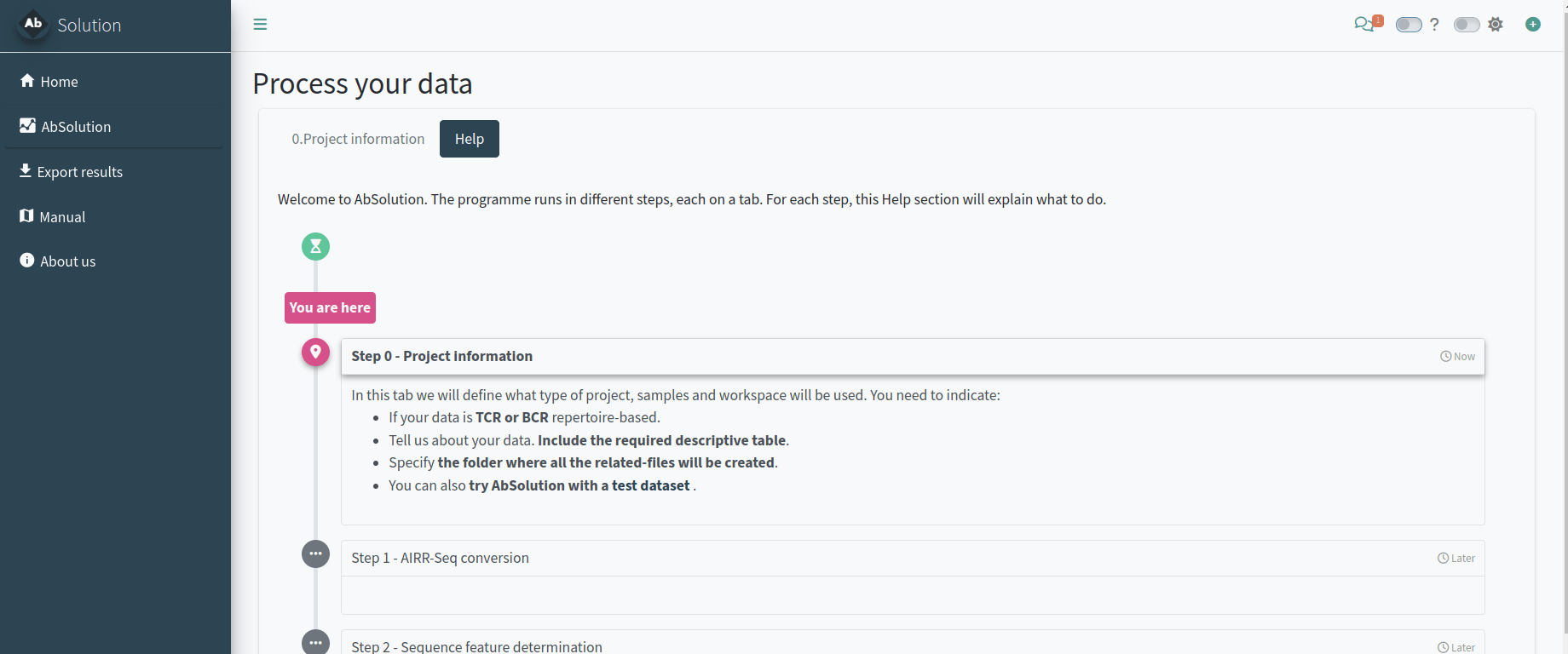
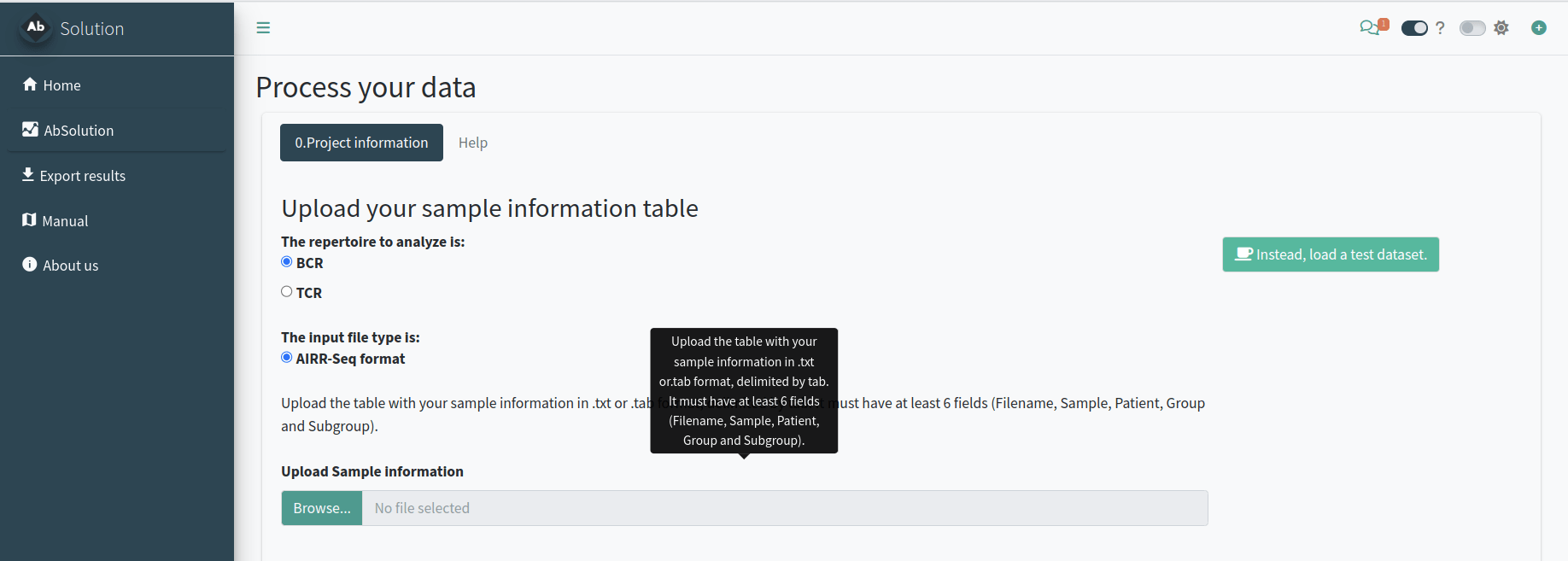
Setting up the project analysis - Step 0
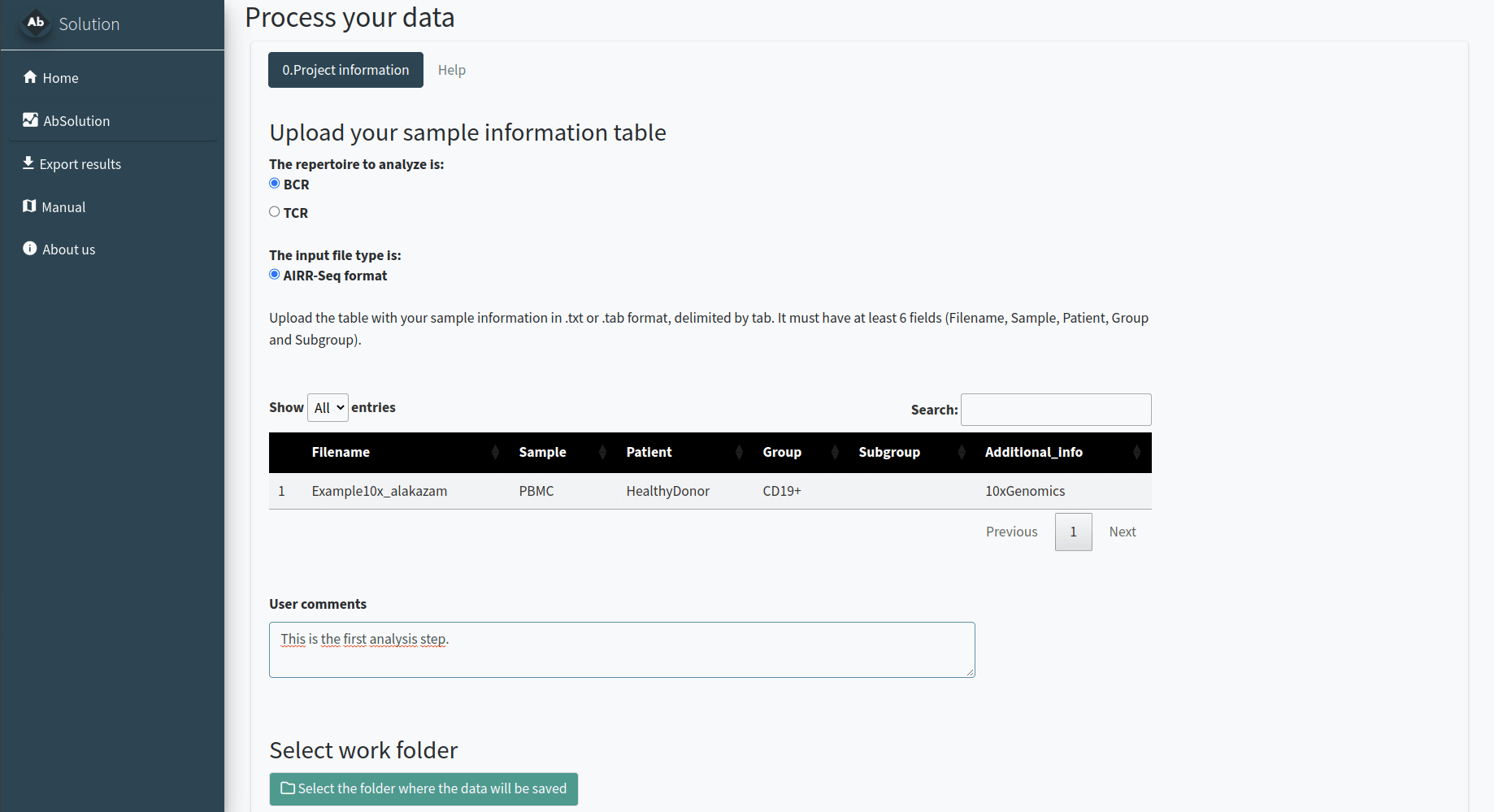
In the 0.Project information tab, we will define the type of project, samples, and workspace to be used. You need to indicate:
- If your data is TCR or BCR repertoire-based.
- Provide details about your data, including the required descriptive table.
- Specify the folder where all related files will be created.
OPTIONAL: try the test dataset
You can try AbSolution with a test
dataset. This is a small, down-sampled dataset of 10x
Genomics Ig V(D)J sequences from CD19+ B cells isolated from the PBMCs
of a healthy human donor. The data, provided by 10x Genomics under a
Creative Commons Attribution license, has been processed using their
Cell Ranger pipeline. It comes from the Alakazam package.

Just click the Instead, load a test dataset button and continue the workflow.
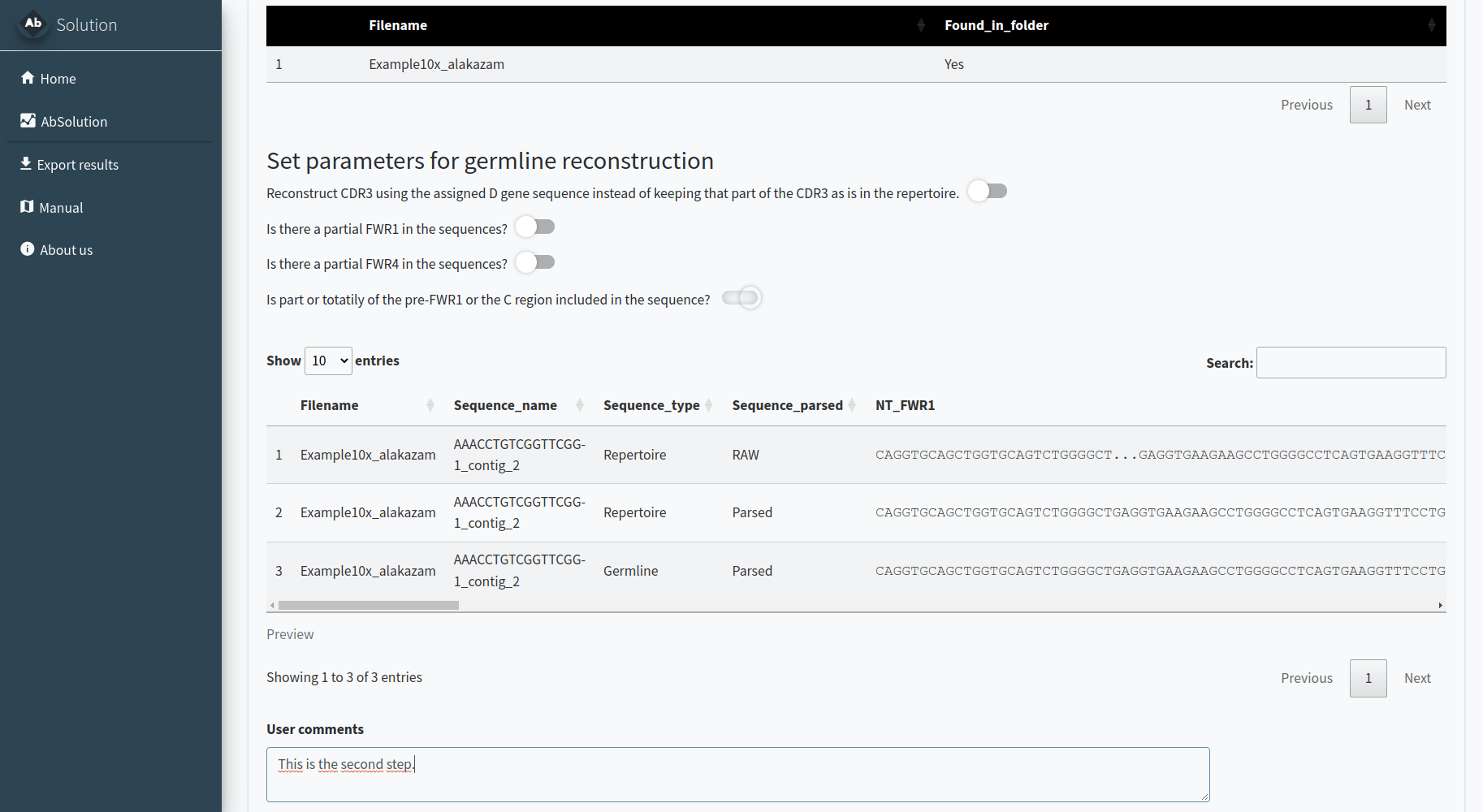
Parsing the sequences - Step 1
In the 1.AIRR-Seq conversion tab, we will reconstruct the
repertoire germlines based on your input. AbSolution works
with full Fab sequences or sequences with partially-sequenced FWR1/4.
You need to indicate:
- The folder where your data is located. All files must be in the same folder. Filenames should match those indicated in the previous step. If they are found in the folder, it will say ‘Yes’ in Found_in_folder; otherwise, it will say ‘No’.
- Additional information to better reconstruct the germlines. Decide whether to keep the CDR3 D-gene segment as is or use the D gene information to reconstruct it. Indicate if the FWR1 or FWR4 are partially sequenced and if there is a pre or post-Fab sequence to be removed.
Please confirm with the pre-visualization that the sequences are correctly parsed and reconstructed. A lowercase letter in the repertoire sequence indicates an insertion, while a lowercase letter in the reconstructed germline indicates a deletion in the repertoire.
Calculating sequence-based features - Step 2
In the 2.Sequence feature determination tab, we will calculate the features (physicochemical, composition, etc.) at the nucleotide (NT) and amino acid (AA) levels for the entire sequence (repertoire and reconstructed germline) and its individual regions.
Just click the button, and wait until it is completed. It can take quite some time depending on your dataset size!
ALTERNATIVE: start with previously calculated files - Skip steps 1 & 2
For pre-calculated data, in step 0 you can select to skip steps 1 and 2. In the 1&2.Select your FBM and associated files tab, you can load the files and proceed directly to step 3.
You need to specify the folder where your pre-calculated FBM files are located. All files must be in the same folder, and filenames should match those indicated in the previous step. If the files are found in the folder, it will display ‘Yes’ in the Found_in_folder column; otherwise, it will display ‘No’.
You can also generate dummy datasets with the same number of mutations to use as a negative reference in the next steps.
Exploring the dataset and filtering sequences and variables - Step 3
In the 3.Dataset exploration and variable selection tab, you can customize your analysis by selecting specific sequences, variables, and groups, ensuring a tailored and comprehensive exploration of your dataset. Variables with NAs, infinite values, or zero variance are automatically removed.
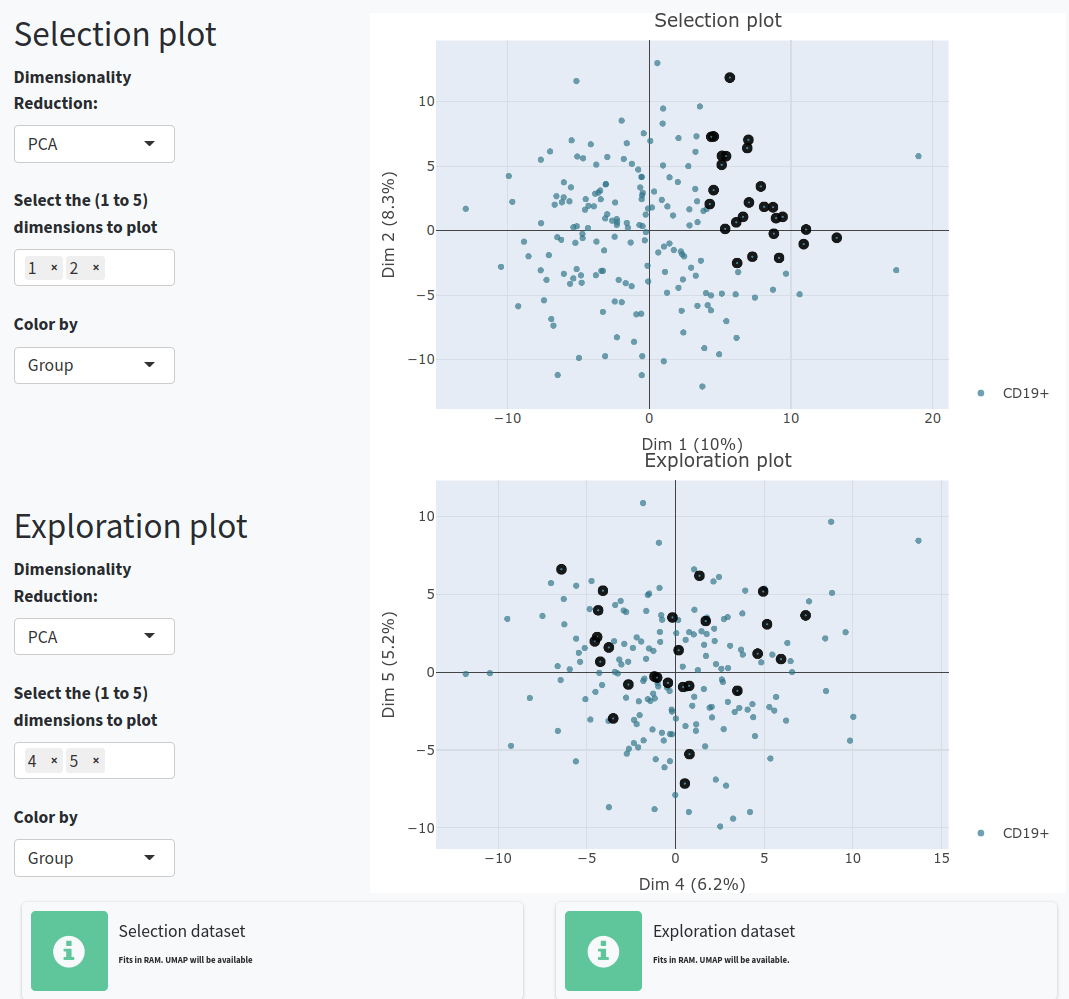
There are different steps and options inside:
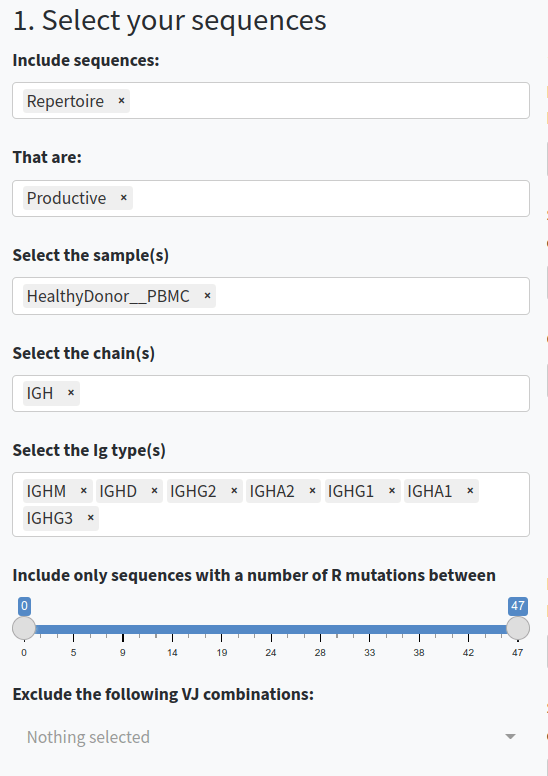
- Select and filter the sequences. You can choose whether to include repertoire sequences, reconstructed germline sequences, or both. You can also filter sequences based on productivity, sample, chain type, and the number of replacement (R) mutations.
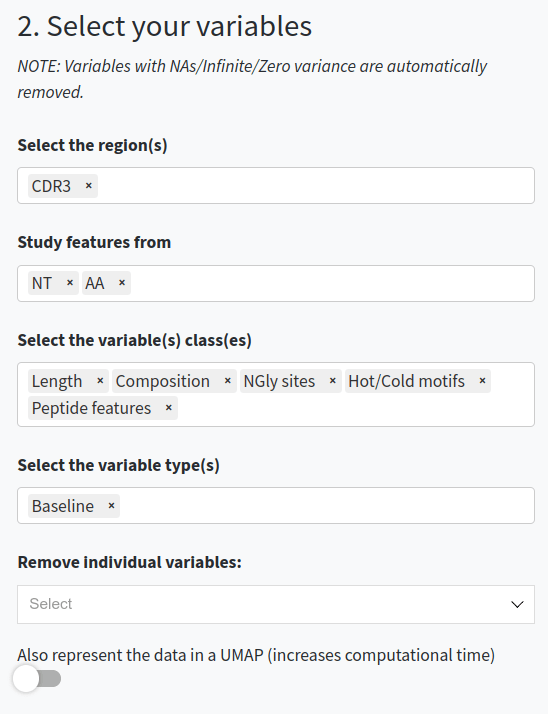
- Select and filter the variables. You can choose which regions (e.g., CDR3, FWR1, Whole Fab), types of sequences-based features (e.g., AA-based, NT-based, composition, physicochemical, etc.) to include in the analysis, or exclude specific features. You can also work with the baseline values or with the germline-difference values.
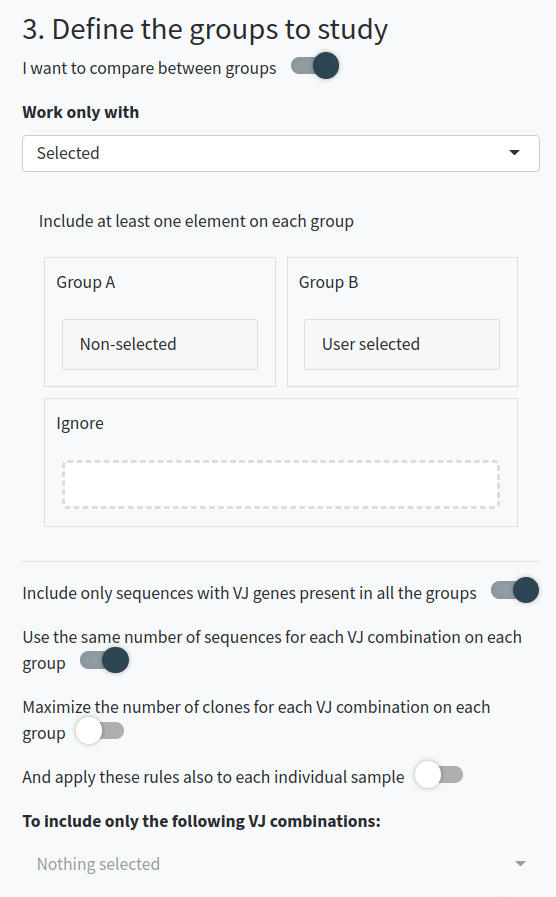
- Decide if you want to compare between groups of sequences. If so, specify which sequences and the type of comparison you want to perform. You can specify groups of sequences for comparison and normalize groups per VDJ usage or clonal usage to avoid overrepresentation. You can also apply these rules to individual samples instead of at a group-level and include only features correlated to the groups.
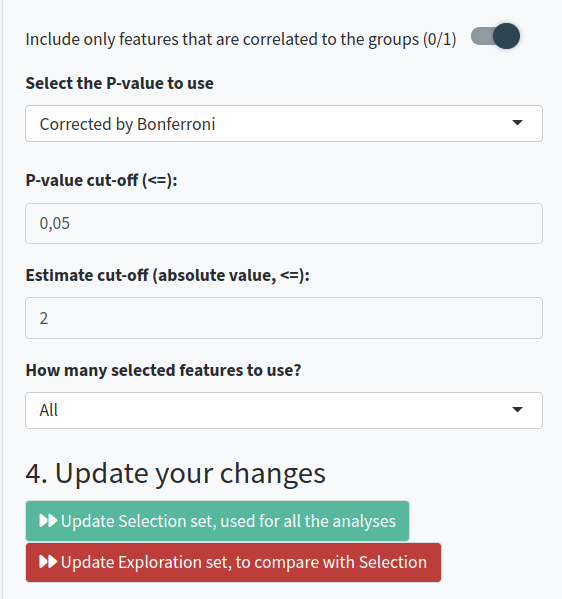
- Perform calculations for the Selection set (which will be shown in the Feature Exploration and Clonal Exploration tabs) or the Exploration set (which you can use to try combinations and compare with the Selection set).
Exploring the variables - Step 4
In the 4.Feature exploration tab, you can plot the filtered variables as a violin plot to observe their behavior according to the selected group in field 4 of the Dataset Exploration and Variable Selection tab. You can add the values as dots, select those of interest, and include the reconstructed germline sequences (if they are not already included) to compare how these values have changed since the germline sequence.
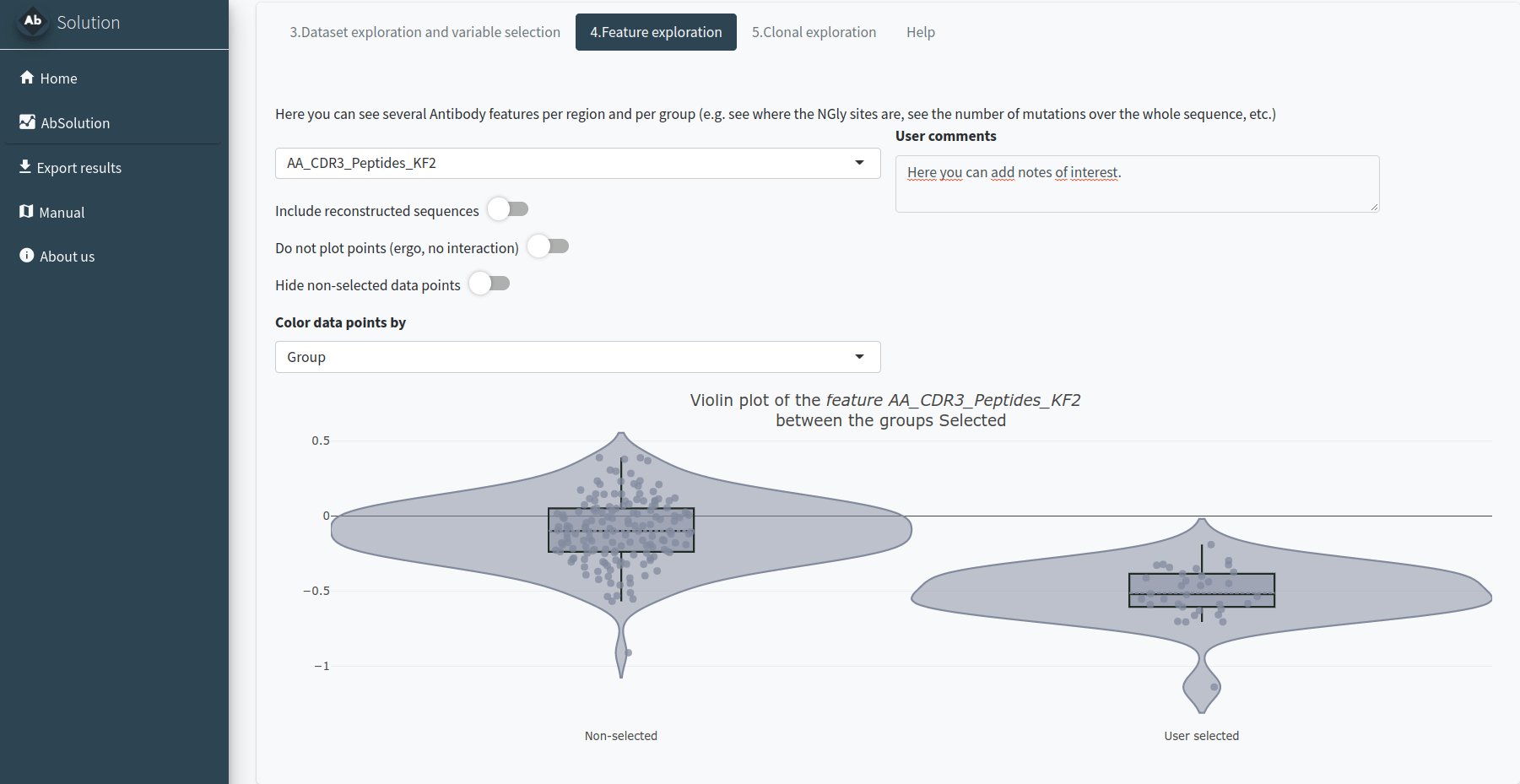
Exploring the clones - Step 5
In the 5.Clonal exploration tab, we will define the clonal definition to be used. You can:
- Use a pre-existing clone definition (Clone_ID) or calculate it de novo.
- Select a dominance threshold based on relative abundance.
- Identify shared clones between samples.
- Manually select clones of interest.
If you have a clonal definition assigned and the images in the clonal and feature tabs have loaded, you will be able to export results in the Export Results tab.
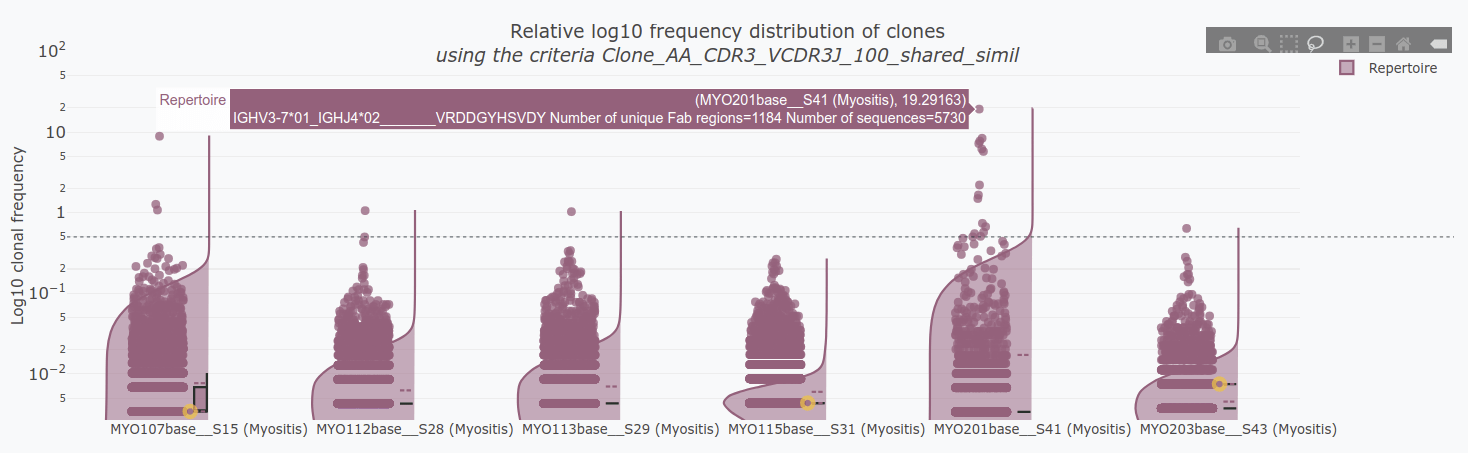
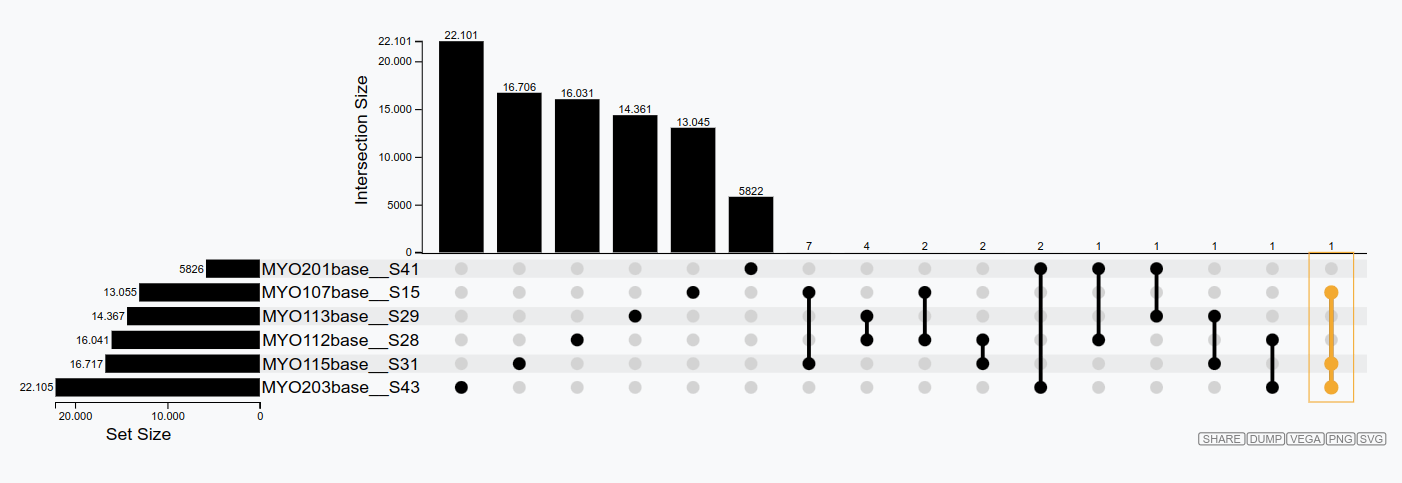
Reproducibility of AbSolution
In the Export results tab, after running a full analysis with the app (this means you need to have generated clonal figures in Step 5), you can download a .zip file that follows the ENCORE structure. This .zip file contains the following subfolders:
-
/0_SoftwareEnvironment/R/: Contains the
AbSolutionpackage in the version used for the analysis, a Docker file, and the renv information to reproduce everything. - /Data/Dataset/: Contains the dataset used for the analysis.
- /Raw/: Contains the sample input files.
- /Meta/: Contains a file with information about the samples.
-
/Processed/: Contains the files produced during the
analysis with
AbSolution(sequence and feature information). These files are used for analysis and data exploration. -
/Notebook/: Contains the .Rmd file to generate the
figures. This .Rmd file is generated directly from the app’s code,
capturing the domain logic using shinymeta. Additionally, once exported,
it uses relative paths instead of absolute paths, making it easy to knit
successfully. The chunks
parse_inputandfeature_calculationare not evaluated unless the user setseval=TRUE, as they are used to produce the Processed files. For time efficiency, this part is skipped when producing the .zip. There are various fields along the pipeline where users can take notes. These notes will be included in the subsequent notebook and results. -
/Results/: Contains the HTML file produced from the
.Rmd file with the key plots from
AbSolution. The HTML file includes all the system information and package versioning details.
It can take some time to load and some time to generate this .zip file. Be patient!